Biochemistry 1 – Flashcards
Unlock all answers in this set
Unlock answersquestion
Polymers of AMINO ACIDS.
answer
Proteins
question
another name for AMINO ACIDS
answer
Residues
question
VERY SMALL chain of amino acids
answer
Oligopeptide
question
Longer chain of amino acids
answer
Polypeptide
question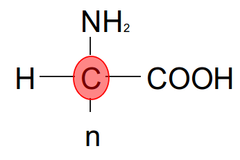
All human amino acids are chiral EXCEPT for GLYCINE. They have an R group, a hydrogen, a Carboxylic acid, and an amine.

answer
Alpha Carbon Stereocenter
question
L or D designations are given based on which side of the Fischer Projection an Amine group is. ALL HUMAN AMINO ACIDS ARE L as they are derived from L-Glyceraldehyde All amino acids with the exception of Cysteine (R) and Glycine have an S stereocenter
answer
Absolute Configuration
question
Depends on the existence of complementary charges (combining in such a way as to enhance or emphasize the qualities of another-in this case, enhancing the chances of binding and stability) Also depends on the hydrophilicity or basicity
answer
Will a substrate bind in an active site?
question
Hydrophobic -R groups fold INTO the protein core (hydrophobic environment) Hydrophilic -R groups are more common on the surface of the protein (hydrophilic environment)
answer
How will a protein fold?
question
what amino acids are present and what is the chemistry of their -R groups?
answer
When you see PROTEIN or ENZYME think:
question
pKa tells us how acidic (or not) a given hydrogen atom in a molecule is. pH tells us how acidic a solution is. For ex: remember in a titration curve how the pH is the dependent variable (measures the pH of the solution)
answer
pH vs pKa
question
pKa ~ 2
answer
Pka estimates: alpha -COOH group
question
pKA ~ 4 (Asp = 3.7, Glu = 4.5)
answer
Pka estimates: -R Group, ACIDIC
question
pKa ~ 6
answer
Pka estimates: -R Group, His
question
pKa ~ 9
answer
Pka estimates: alpha -NH3+ group
question
pKA ~ 11-12 (Lys = 10.7, Arg = 12)
answer
Pka estimates: -R group, BASIC
question
TAKE TABLETS. Your body CANNOT synthesize this amino acid. You must INGEST it.
answer
Essential amino acids
question
your body can SYNTHESIZE this amino acid on its own.
answer
Non-essential amino acids
question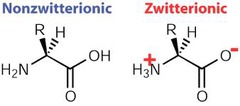
dipolar version of an amino acid wherein positively and negatively charged functional groups cancel one another out, resulting in a neutral ion.

answer
Zwitterion
question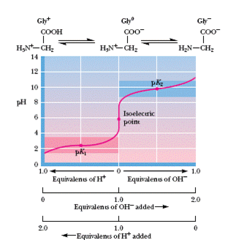
pH at which a molecule carries no Net Charge (often a zwitterion). For a molecule with functional groups having different pkas: pI = (pKa1 + pKa2)/2

answer
Isoelectric point (pI)
question
average of pKa amine group and pKa carboxyl group
answer
pI neutral
question
average of pKa ACIDIC R group and pKa carboxyl group
answer
pI acidic
question
average of pKa amine group and pKa BASIC R group
answer
pI basic
question
where [titrant] = [anylate] acid and base are present in equal quantities (neutralized). The solution is neutral.
answer
Equivalence Point
question
At half equivalence point, [HA] = [A-], therefore pH = pKA The amount of acid/base and it's conjugate are of equal concentration.
answer
Half Equivalence Point
question
point where the indicator causes a color change to help determine the equivalence point. Common Indicators by pH: Phenylphthalein: 9.3 Litmus: 6.5 Methyl Orange: 3.7
answer
End Point
question
The region of a titration curve in which the concentration of a conjugate acid is approximately equal to that of the corresponding base. The pH remains relatively constant when small amounts of H+ or OH- are added because of the combination of these ions with the buffer species already in solution. FLAT portion of a titration curve
answer
Buffer Region
question
occurs via Dehydration synthesis and acyl substitution. The amine group nitrogen (nucleophile) from the New amino acid attacks the carbonyl carbon (electrophile) of the C-TERMINUS of the growing peptide chain (aided by enzymatic function of the ribosome).
answer
Peptide bond formation
question
peptides are WRITTEN, READ, and SYNTHESIZED from N-TERMINUS --> C-TERMINUS
answer
Peptide Chain Conventions
question
resonance between pi electrons of the C=O bond, and the nitrogen pair of the C-N bond, yield 2 resonance structures for any peptide bond. THE ACTUAL STRUCTURE IS A HYBRID of the two. BOTH THE C=O BOND AND THE C-N BOND IN A PEPTIDE BOND HAVE DOUBLE BOND CHARACTER (rigid peptide bond with limited rotation).
answer
Resonance between peptide bonds
question
cleaves ARGININE and LYSINE on the CARBOXYLIC SIDE think of the one with extra amino groups
answer
Trypsin (protein hydrolysis)
question
cleaves PHENYLALANINE, TRYPTOPHAN, and TYROSINE on the CARBOXYLIC side. think of the R-Groups with aromatic rings It is made of Serine, Histidine, and Aspartate
answer
Chymotrypsin (protein hydrolysis)
question
AA-AA-AA-AA-AA The amino acid sequence Formation of Covalent Peptide Bonds
answer
Primary Protein Structure
question
Alpha Helices and Beta Pleated Sheets Primarily the result of Hydrogen Bonding
answer
Secondary Protein Structure
question
Hydrogen bonding between CARBONYNL OXYGENS and AMIDE HYDROGENS that are exactly FOUR resides apart. ONLY every FOURTH residue is involved in hydrogen bonding R groups are directed exactly away from the alpha helix cylinder (i.e. perpendicular to a plane tangent to the surface of the alpha helix)
answer
Alpha Helices
question
Hydrogen bonding between ALL of the CARBONYL OXYGENS in one row and the AMIDE HYDROGENS in the adjacent row. ALL residues are involved in hydrogen bonding R groups are directed perpendicular to the plane of the beta sheet, on both sides. Beta sheets are in a PLEATED conformation. This is necessary for the carbonyl and amide moties to line up properly so that EVERY RESIDUE is participating in TWO HYDROGEN BONDS.
answer
Beta Pleated Sheets
question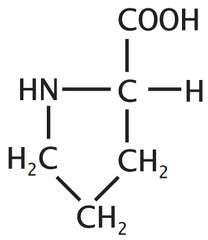
Usually the first residue at the very end of an alpha helix, but rarely found inside the helix because it introduces KINK/TURN. This same KINK/TURN is desirable at the end of beta-sheets because the chain must make a 180 degree turn to align as a neighboring row in a beta sheet. (SEE TOP end of Beta Sheet)

answer
Proline
question
found in hair and nails = alpha helices
answer
Common applications of secondary Structures: Keratin
question
molecule that makes up silk = beta sheets
answer
Common applications of secondary Structures: Fibroin
question
Geometric, three-dimensional folding of the alpha helices, beta sheets, and other moieties to form a functional globular or structural protein.

answer
Tertiary Protein Structure
question
1) HYDROGEN BONDING between N-H and C=O or side chains (amine, carboxyl, and alcohol groups) 2) DISULFIDE BONDS - covalent bond between 2 cysteine residues 3) HYDROPHILIC/HYDROPHOBIC INTERACTIONS- in soluble proteins, hydrophobic amino acids collapse into the protein core. In membrane proteins, hydrophilic membranes will be outside the membrane in the cytoplasm or in the core of the protein. Hydrophobic amino acids will be located in the bilayer. 4) IONIC INTERACTIONS - charge-charge interactions between a positively charged amino acid (ex: lysine) and a negatively charged amino acid (aspartic acid) (NH3+ R group interacting with COO- R group - not the zwitterion part) 5) VAN DER WAALS FORCES - intermolecular forces that repel atoms away from each other (steric hindrance) 6) PROLINE TURNS - proline's unusual shape will cause a KINK in the middle of an alpha helix, but it aids in beta turns (see proline)
answer
6 Molecular interactions that contribute to a tertiary protein structure
question
association of multiple folded proteins into a multi-subunit complex: Ex: Hemoglobin
answer
Quaternary Protein Structure
question
consists of 4 protein chains (2 alpha and 2 beta subunits) - quaternary structure. Each subuint has one heme group capable of binding 1 O2 molecule so 1 hemoglobin binds 4 O2 molecules
answer
Hemoglobin
question
LIGAND AFFINITY INCREASES WITH THE BINDING OF EACH SUBSEQUENT LIGAND. In hemoglobin, affinity of the first oxygen is relatively low, but it increases for the second, third, and fourth oxygen to bind. This affinity remains in effect during offloading of oxygen at the tissues. Therefore, the first oxygen (highest cooperative affinity) dissociates at the slowest rate, but each subsequent oxygen is released more easily.
answer
Positive Cooperativity
question
Globular proteins are SPHERICAL proteins that are somewhat water-soluble (they form colloids in water), unlike fibrous or membrane proteins.
answer
Globular protein
question
a translated protein assumes secondary structure almost immediately, and then folds into its globular or structural tertiary state
answer
Protein folding
question
FULLY FOLDED protein
answer
Globule
question
PARTIALLY folded protein
answer
Molten globule
question
FULLY UNFOLDED protein; a.k.a DENATURED protein
answer
Molten
question
hydrophobic -R groups fold into the INTERIOR of a globular protein to escape water. They often bring some small polar groups with them, which interact in a complementary way to stabilize the folded protein further. Charged molecules can be found in a hydrophobic core if they are paired with a complementary side chain of the opposite charge
answer
Hydrophobic core (protein folding interactions)
question
the majority of -R groups on the surface of a globular protein are either polar or charged
answer
Hydrophilic surface (protein folding interactions)
question
interactions between charged-R groups both encourage the act of folding itself, and stabilize the protein in its folded state
answer
Electrostatic Interactions (protein folding interactions)
question
hydrogen bonding between -R groups also encourages folding and stabilizes the folded protein
answer
Hydrogen Bonds (protein folding interactions)
question
two oxidized cysteine residues form a disulfide (R-S-S-R) bond. This is the STRONGEST TYPE OF PROTEIN FOLDING interaction. Disulfide bonds between keratin alpha helices are what make hair more or less curly.
answer
Disulfide bonds (protein folding interactions)
question
formed when acidic and basic -R groups undergo a neutralization reaction resulting in a salt
answer
Salt Bridges (protein folding interactions)
question
can be considered as either DISRUPTING secondary structure or as CONTRIBUTING to the tertiary structure. Neither alpha helices, nor beta sheets can contain proline internally without disruption of the secondary structure. However, proline residues are often found at the beginning of alpha helices and are very common (along with glycine) in the sharp turns at the end of two adjacent rows in a beta sheet.
answer
Proline Turns (protein folding interactions)
question
a layer of water that surrounds a dissolved protein. The water molecules in this layer interact closely with each other and with the protein's surface. The water in the hydration layer is more ordered than the bulk water in the general area and is considered not to participate with the bulk (a.k.a. unstructured) water when considering colligative (relating to the binding together of molecules) properties
answer
Solvation Layer (protein folding interactions)
question
It is true that entropy of a protein decreases when it is folded (more ordered). The surrounding also matters however (part of the system). In an unfolded protein, hydrophobic portions of the polypeptide are exposed to water molecules which order themselves into hydration spheres (ordered). When a protein is folded, these hydration spheres break down (entropy of water molecules/surrounding increases). In other words, while entropy of the protein decreases (more stable) due to folding, the entropy of the surrounding region of water increases, leading to an OVERALL increase in entropy. Nonpolar solvation (very ordered water molecules) --> Polar/charged solvation (less ordered water molecules). Favorable INCREASE IN ENTROPY is a major CONTRIBUTOR to the OVERALL CONFORMATIONAL STABILITY of the folded protein. Increase in entropy is proportional to the conformational stability of a protein.
answer
Entropy and Protein folding (protein folding interactions)
question
-acid-pH (destroys tertiary and quaternary structure) -Chemicals (destroys secondary, tertiary, and quaternary structures) -Temperature (destroys secondary, tertiary, and quaternary structures) -Enzymes (destroys primary, secondary, tertiary, and quaternary structures)
answer
Protein Denaturing Agents
question
Isoelectric focusing is separating proteins by isoelectric point (pI). Proteins are placed in a gel with a stable pH gradient. If a protein is in a region of the gel with a lower pH than its pI, then the protein will be POSITIVELY charged and move towards the negative cathode. If a protein has a pH higher than its pI, it will be NEGATIVELY charged and move towards the positive anode. As the proteins move from their pHs to their respective pIs, their charges NEUTRALIZE and the protein will eventually stop to move (no charge, so no more pull towards either direction).
answer
Protein separation technique: Isoelectric point (pI)
question
Electrophoresis separates proteins based on size. Proteins are first placed in a detergent which DENATURES them and coats them with a NEGATIVE charge. This gives proteins a UNIFORM charge/mass ratio. Proteins are then run through a gel of polyacrylamide which slows down the migration of large proteins over the smaller ones. Gel is run from - to + electrodes and so the proteins are pulled towards the + electrode. The smallest proteins will run furthest through the gel, while the largest proteins will stay near the top.
answer
Protein separation technique: Electrophoresis
question
DNA --> RNA --> Proteins DNA and RNA has NUCLEIC ACID building blocks Proteins have AMINO ACID building blocks
answer
Central Dogma
question
Hemoglobin, Calmodulin, Troponin, Tropomyosin, Histones, Transcription factors, Cell adhesion molecules
answer
Common Binding Proteins
question
Antigens (foreign bodies), Antibodies (produced in the body to fight off antigens)
answer
Immune System Proteins
question
Actin (thin filaments, microfilaments), Tubulin (microtubules), Keratin (hair and nails, intermediate filaments), Elastin (connective tissue, extracellular matrix)
answer
Structural Proteins
question
Myosin (power stroke, cellular transport), Kinesis and Dyneins (vesicles, cellular transport, cell division, cilia, flagella)
answer
Motor Proteins
question
Move along microtubules from (CENTER OF CELL to PERIPHERY; NERVE CELL BODY --> DENDRITE)
answer
Kinesins
question
Move alone microtubules (+) to (-) end (PERIPHERY to CENTER OF THE CELL; nerve cell DENDRITE --> CELL BODY) Think Dyenin so D for dendrite
answer
Dyneins
question
PROTEINS increase reaction rate, lower activation energy, catalyze both forward and reverse reactions, NOT consumed in the process, NEVER alter thermodynamic properties of a reaction, and MOST NOTABLE DIFFERENCE: As proteins, enzymes are far more sensitive than inorganic catalysts to environmental conditions such as temperature and pH.
answer
Enzymes
question
REDOX reactions
answer
Oxidoreductases (enzyme classification by reaction type):
question
Transfer of a functional group (e.g. kinases, aminotransferases)
answer
Transferases
question
Hydrolysis (cleavage with H2O)
answer
Hydrolases
question
Rearrangements (e.g. phosphoglucose isomerase [G6P-->F6P], epimerases)
answer
Isomerases
question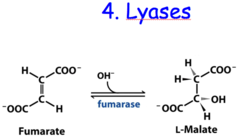
AB A + B [cleavage/synthesis; No H2O, not hydrolysis]

answer
Lyases
question
Addition or synthesis of LARGE molecules, usually ATP DEPENDENT (e.g. DNA Ligase)
answer
Ligases
question
enzymes help reactions Over The HILL (capital letters)
answer
Enzyme Classification Mnemonic:
question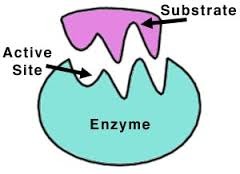
A substance acted upon by an enzyme.

answer
Substrate
question
place on the enzyme where the substrate binds.
answer
Active Site
question
Enzyme substrate complex forms when a substrate is bound to the active site and converted into a product.
answer
Enzyme-Substrate Complex
question
E + S ES --> EP E + P
answer
Basic Kinetics Formula
question
The active site's empty structure is not an exact fit for the substrate. As the substrate begins to bind the active site pocket, small but specific conformational changes occur . This is FAVORED as when the substrate binds, the affinity for the substrate increases. The resulting conformational changes induce a higher affinity for the transition state by stabilizing it and lowering the activation energy.
answer
Induced Fit Theory
question
predicts that the ACTIVE SITE IS AN EXACT FIT for its substrate. This theory is NOT FAVORED by scientists because it predicts a rigid, inflexible active site.
answer
Lock and Key Theory
question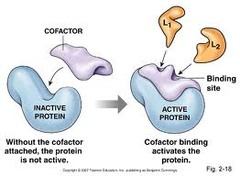
general term for any species required by an enzyme to function; coenzymes and prosthetic groups are both examples of cofactors

answer
Cofactors
question
Non-Protein species TEMPORARILY attached to the enzyme but REQUIRED by the enzyme to function
answer
Coenzymes
question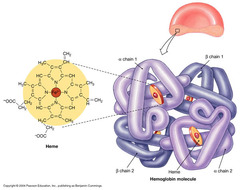
Non-Protein groups that ARE PERMANENTLY attached to the enzyme and are required by the enzyme to function. Ex: Iron in Hemoglobin

answer
Prosthetic groups
question
protein that contains ONLY AMINO ACIDS and no non-protein cofactors or prosthetic groups.
answer
Simple Protein
question
protein that is assoicated with its cofactors, either covalently or via intermolecular attractions. Hemoglobin is a conjugated protein because it contains the non-protein heme group (Fe2+ and 4 pyrrolic groups attacked to it).
answer
Conjugated Protein
question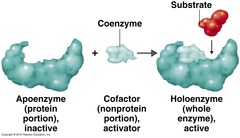
SIMPLE PROTEIN that is an ENZYME

answer
Apoenzyme
question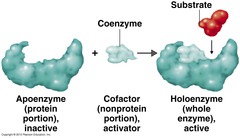
Apoenzyme + cofactor. It is ready to bind to a substrate. CONJUGATED PROTEIN that is an ENZYME.

answer
Holoenzyme
question
ADEK Stored in the liver and adipose tissue. Can lead to hypervitaminosis (toxic levels of vitamins) Antioxidants (vitamin E) Bloodclotting/blood pressure (vitamin K) Eyesight (Vitamin A) Calcium Levels (Vitamin D)
answer
Fat Soluble Vitamins
question
All of the rest Water soluble vitamins act as cofactors
answer
Water Soluble Vitamins
question
small, ORGANIC molecules, essential nutrients necessary for metabolism. Humans have lost the ability to synthesize vitamins in sufficient quantities. We need vitamins in trace amounts so it takes less energy to obtain from DIET than to synthesize on our own. Vitamins have to be modified later in the body.
answer
Vitamins
question
small, INORGANIC molecules, necessary for bone formation (calcium and phosphate), ion gradients (sodium and potassium), oxygen transport (iron containing heme group), muscle contraction (calcium), ATP processing (magnesium), stomach acid production (chlorine), etc. Obtained via DIET. Needed in small quantities (MICRONUTRIENTS)
answer
Minerals
question
E + S ES --> E + P
answer
Steady State assumpuption Kinetics Formula:
question
V = (Vmax*[S])/(Km+[S]) The relationship between REACTION VELOCITY, Km, and SUBSTRATE CONCENTRATION
answer
Michaelis Menten Equation
question
the theoretical maximum rate of a reaction where an enzyme and substrate bind. At low [S], reaction rate increases rapidly because there is a higher chance for the free substrates to encounter an empty enzyme. As [S] increases, , the likelihood of encountering empty enzymes by free substrates decreases. At Vmax, all enzymes are bound to (preoccupied with) a substrate and as soon as a substrate is converted into a product, another substrate binds to the enzyme.
answer
Vmax
question
When all of the enzymes are preoccupied with substrates, the reaction is deemed SATURATED
answer
Saturated
question
Km IS THE RELATIVE MEASURE OF AN ENZYME'S AFFINITY FOR ITS SUBSTRATED (Km = (k-1 + k2)/k1 Km = [S] @ Vmax/2
answer
Michaelis Constant: (Km)
question
a double-interse graph of the reaction rate (v inverted to 1/v) and substrate concentration ([S] inverted to 1/[S]) Applications: Used to determine what kind of enzyme inhibition is occurring as we can easily calculate the IMPACT of an INHIBITOR on the Km and Vmax.
answer
Lineweaver-Burk Plots
question
Inhibitor is not permanently bound to the enzyme; enzyme is not completely disabled
answer
Reversible Enzyme Inhibition
question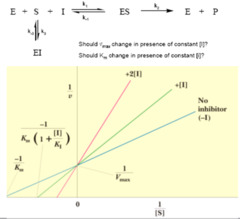
INHIBITOR BINDS AT THE ACTIVE SITE. The inhibitor resembles the substrate in shape; the inhibitory effect CAN BE OVERCOME by INCREASING the CONCENTRATION OF SUBSTRATE Vmax = No Change Km = Increases Ex: Statin is a competitive inhibitor which binds to the active site of the HMG-CoA reductase enzyme and inhibits the enzyme's function of producing cholesterol in the liver. Chelators bind to the active site in order to be efficient

answer
Competitive Inhibition
question
Inhibitor binds ONLY WITH THE ENZYME-SUBSTRATE COMPLEX. Typically RARE. Binds to another site which opens up when the ES complex is formed. Vmax = Decreases Km = Decreases Ex: Lithium is a drug used to treat maniac depression. Lithium acts as an uncompetitive inhibitor in the phosphoinositide pathway, inhibiting inositol monophosphate and preventing the inositol recycling in the brain. (inositol recycling is probably what is leading to the maniac depression)
answer
Uncompetitive Inhibition
question
INHIBITOR BINDS AWAY FROM THE ACTIVE SITE and changes the shape of the enzyme. The inhibitor has an EQUAL affinity for both the enzyme-substrate complex (E-S) and the enzyme (E). Vmax = Decreases Km = No Change Ex: Alanine acts as a non-competitive inhibitor for the enzyme pyruvate kinase. Pyruvate kinase normally transfers one phosphate group from PEP to ADP, creating pyruvate and one ATP. Alanine can bind to pyruvate kinase (whether or not PEP) the substrate is bound to inhibit activity. This creates a negative feedback loop since alanine syntheiss is derived from pyruvate.
answer
Non-Competitive Inhibition
question
Inhibitor has UNEQUAL affinity for the E-S and the E, favoring one over the other. Vmax = Decreases Km = Decreases if Inhibitor binds to E-S over E Km = Increases if Inhibitor binds to E over E-S Ex: Metal palladium is a mixed inhibitor for xanthine oxidiase (enzyme that prevents xanthine to convert into uric acid.
answer
Mixed Inhibition
question
Inhibitor binds COVALENTLY to the enzyme and/or the active site, DISABLING the ENZYME for either a prolonged period of time, or PERMANENTLY. Ex: Aspirin is an irreversible inhibitor which binds to cyclooxygenase 1 and 2 irreversibly to prevent their inflammatory responses.
answer
Irreversible Inhibition
question
a specific type of non-competitive or allosteric inhibition that applies to multi-step reactions, synthetic pathways, or cascades. ONE OF THE PRODUCTS OF A REACTION LATER IN THE CHAIN ACTS AS AN INHIBITOR FOR ONE OF THE ENZYMES EARLIER IN THE CHAIN.
answer
Feedback Inhibition (a.k.a NEGATIVE FEEDBACK)
question
product of a reaction acts as an AGONIST for the reaction. Ex: Product is an activator so more product = more reaction. In blood coagulation, upon damage to the blood vessel, glycoprotein FVII is released. An activation cascade of coagulation occurs resulting in prothrombin being activated to thrombin and throbin activating more FVII. This results in an INCREASE in the cascade happening.
answer
Positive feedback
question
Inactive enzyme precursor. Zymogens prevent enzyme activity during MANUFACTURE and TRANSPORT. Ex: prothrombin in converted to its activated form thrombin. It is better to have enzymes in their inactive states because they can be detrimental if they are active all the time. Imagine having activated proteases all the time. They will degrade cells continuously and that can be bad
answer
Zymogen
question
enzymes whose activity is influenced by the reversible, non-covalent, bonding of another molecule (i.e. second subunit or an activating/deactivating molecule). Ex: Threonine deaminase is an allosteric enzyme. It has 2 binding sites. When isoleucine binds to the high affinity site, binding affinity for the low affinity site increases (something else will bind there to activate it). If isoleucine binds to the low affinity site, the enzyme deactivates.
answer
Allosteric enzymes
question
if something ends with -ase (ex: synthase), it is an enzyme. If something ends with -tase (ex: synthetase), it is an enzyme which uses energy (like ATP). Be careful because some enzymes which use energy can apply to reversible reactions and even though energy is used in going only one way, it still has the -tase ending.
answer
-ase, -tase hints
question
Empirical formula of all monosaccharides = (CH2O)n Empirical formula of all polysaccharides = Cn(H2O)x
answer
Carbohydrate Empirical Formulas
question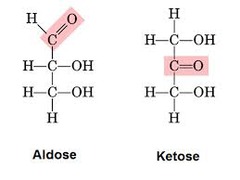
-"-ose" ending is given to all sugars (i.e. glucOSE, ribOSE) -"deoxy-" prefix is used in the normal location of an -OH group is replaced with Hydrogen (i.e. DNA vs. RNA) Aldose vs. Ketose (aldehyde vs. ketone)

answer
Carbohydrate Nomenclature and Classification
question

answer
Glyceraldehyde vs. Dihydroxyacetone
question
Alpha glucose vs. beta glucose (ANOMERS) Animals have ALPHA glucose (OH on 1 C down) Plants have BETA glucose (OH on 1 C up) monosaccharide -OH groups on glucose can function as NUCLEOPHILES
answer
Glucose
question
Linear form is a ketose monosaccharide
answer
Fructose
question
monosaccharide
answer
Galactose
question
monosaccharide

answer
Mannose
question
Galactose + Glucose (beta linked by 1,4-glycosidic linkage) disaccharide
answer
Lactose
question
Glucose + Glucose Think single malt and double malt disaccharide
answer
Maltose
question
Glucose + Fructose Think of coke in US (uses fructose) vs. coke in mexico (uses sucrose) disaccharide
answer
Sucrose
question
Pyranose (6 member ring) Furanose (5 member ring). THINK F for Five
answer
Pyranose vs. Furanose
question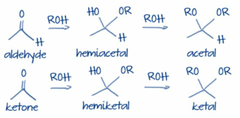
Hemiacetal (cyclical aldehydes) Hemiketals (cylical ketones)

answer
Hemiacetals vs. Hemiketals
question
NOT THE SAME THING! When focusing on R or S, rank the substituents by priority group (atomic number I think) When focusing on D or L, DRAW a FISCHER PROJECTION! If the hydroxyl group attached to the highest numbered chiral carbon is on the right in the Fischer projection, it is D-; if it is on the left it is L-.
answer
R/S vs D/L
question
ALL HUMAN BODY SUGARS are D-Sugars L sugars DO NOT OCCUR naturally in humans
answer
D-Sugars
question
ENANTIOMERS (same molecule, but different stereochemistry) L - furthest -OH group from the CARBONYL is on the LEFT in the Fischer projection D- furthest -OH group from the CARBONYL is on the RIGHT in the Fischer projection Same molecule; different stereochemistry at the last chiral carbon
answer
D-Glucose vs. L-Glucose
question
SAME molecule; different stereochemistry at the anomeric carbon Anomeric carbon = carbonyl carbon in the Fischer projection
answer
Anomers
question
Ex: Glucose vs. Galactose, Mannose vs. Glucose (Fischer projection) DIFFERENT MOLECULES, an example of diastereomers (R vs S configurations are different at one specific chiral carbon) NOTICE that ENANTIOMERS and ANOMERS have the SAME name (glucose in all of these examples), whereas the EPIMERS have DIFFERENT names (glucose/galactose)
answer
Epimers
question
All the OH groups pointing RIGHT on the Fischer projection will point DOWN in the ring structure. All the OH groups pointing LEFT on the Fischer projection will point UP in the ring structing
answer
DownRight UpLefting
question
You can tell a ketose from an aldose in a chain form because of the presence of a ketone functional group instead of an aldehyde functional group (Fischer projection). In ring form, you can tell by looking for R groups on BOTH sides of the ring oxygen. If , as in fructose, you see an R-OH group attached to the carbons on BOTH sides of the ring oxygen, then you know it is a KETOSE. On the other hand, if, as in glucose, ONE side has an R-OH group and the other has ONLY an -OH group, then it must be an ALDOSE.
answer
Aldose vs. Ketose Distinctions
question
INTRAMOLECULAR NUCLEOPHILIC SUBSTITUTION: The -OH group on te chiral caron that is furthest fromt eh carbonyl carbon (the same one used to determine D/L) acts as the nucleophile, attacking the carbonyl carbon (electrophile). The carbonyl oxygen is protonated to form a hydorxyl group.
answer
Carbohydrate Reactions: Ring Closing
question
Using water to break polymers. Polymer (n) + H2O --> Polymer (n-1) + Monomer
answer
Carbohydrate Reactions: Hydrolysis of the Glycoside Linkage
question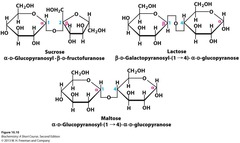
Monosaccharides --> Disaccharides --> Polysaccharides alpha-linkage = linked through an oxygen that is on the OPPOSITE side of the plane from the CH2OH group (i.e. trans) beta-linkage = linked through an oxygen that is on the SAME side of the plane from the CH2OH group (i.e. cis)

answer
Polymerization
question
Glycogen: VERY branched, alpha-linked glucose polymer, used for energy storage in ANIMALS. Starch: branched, alpha-linked glucose polymer, used for energy storage in PLANTS Cellulose: Beta-linked glucose polymer, used for energy storage in PLANTS, indigestible in animals without using symbiotic bacteria (cows). Humans CANNOT digest cellulose.
answer
Glucose polysaccharides
question
Lipids are a class of BIOMOLECULES such as fats, oils, waxes, sterols, fat-soluble vitamins, glycerides (mono-, di-, tri-), phospholipids, and terpenes FOR MCAT Focus: 1) Lipids are BIOMOLECULES 2) Lipids are HYDROPHOBIC
answer
Lipids
question
Fatty acids are the ONLY lipids that have a -COOH group. Usually a long hydrocarbon skeleton with 16-18 carbons with a carboxylic acid attached at the end. AMPHIPATHIC (carboxylic acid is polar)
answer
Fatty Acids
question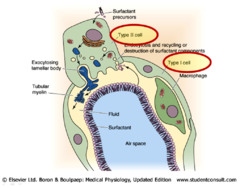
An amphipathic molecule secreted by cells in the alveoli (type II alveolar cells) that reduces surface tension on the inside of the alveolar walls. This prevents the alveoli from collapsing upon exhale and sticking together, thus reducing the effort required for inspiration. Similar to a fatty acid in structure (polar head and non polar tail)

answer
Surfactant
question
Glycerol backbone with 3 fatty acids attached via ESTER linkages NOT Amphipathic
answer
Triacylglycerol (Triglycerides)
question
Saturated fats are saturated with Hydrogen. Saturated fats do not have any double bonds. Unsaturated fats have double bonds (kinks) which can be in either the cis or trans orientation. Unsaturated fat is healthier since it generates fewer calories. Saturated fat has the most stored energy so it is great to consume under famine conditions. Trans-unsaturated fat cannot be broken down in the body and it accumulates in the blood. Trans-unsaturated fat is not created in the body and is ingested via processed foods. Your body can however breakdown ONLY the cis-unsaturated fat. Saturated fat is SOLID at room temperature as they typically have a HIGHER MELTING POINT. Saturated fat can lead to cardiovascular disease.
answer
Saturated vs. Unsaturated fats
question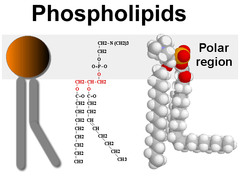
Two fatty acid moieties with a phosphate group that is attached to some other functional group (ex: like choline to make phosphatidycholine). The two fatty acid moieties and the phosphate group are attached to a glycerol backbone. HAS ESTER GROUPS AMPHIPATHIC (phospho group is polar)

answer
Phospholipids
question
The hydrolysis of an ester (base promoted ester hydrolysis used NaOH or something like that). Triglycerides, phospholipids, etc can be hydrolyzed
answer
Saponification
question
Tetracyclic (4) ringed. NOT Amphipathic
answer
Steroids
question
Polymer of ISOPRENES (can be a chain or a ring) A terpene is a repetitive chain of hydrocarbons (usually multiples of 5) Terpenoids - Terpene with OXYGEN NOT Amphiapthic
answer
Terpenes
question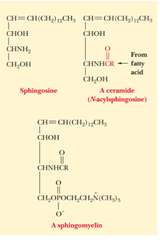
sphingomyelins (phosphate group on 4th carbon on the backbone is attached to choline or ethanolamine, make up the myelin sheath. It's breakdown results multiple sclerosis) or cerebrosides (4th carbon on the backbone is attached to a sugar monomer) AMPHIPATHIC (oxygen and nitrogen are polar)

answer
Sphingolipids
question
Waxes consist of a longchain fatty acid linked through an ester oxygen to a long-chain alcohol. These molecules are completely water-insoluble and generally solid at biological temperatures. Their hydrophobic nature allows them to function as water repellents on leaves of some plants, on feathers, and on the cuticles of certain insects. NOT Amphiathic
answer
Waxes
question
Phosphatid (glycerol backbone that has 2 fatty acid moieties and phosphate group) where the phosphate group is attached to a sugar monomer
answer
Glycolipids
question
Play a key role in inflammation, blood pressure, blood clotting, fever (good when fighting infection), pain, labor, sleep-wake cycle. NOT Amphipathic Lipid mediators that have AUTOCRINE (self-target) and PARACRINE (target = cell in immediate vicinity) functions throughout the body UNLIKE ENDOCRINE HORMONES: -Produced and released throughout the body;NOT only in specialized glands -Act LOCALLY, rather than traveling to a distant target via the bloodstream.
answer
Prostaglandins (PGX)
question
(moles of solute)/(kg. of solvent) Less concentrated than molarity https://www.youtube.com/watch?v=4Btd3FEoI5k
answer
Molality



