Neurochemistry of Behavior: The Sleep-Wake Cycle – Flashcards
Unlock all answers in this set
Unlock answersquestion
Encephalitis lesions affecting the sleep-wake cycle
answer
Shown here is a figure from a paper written by the neurologist Constantin Von Economo. Anterior hypothalamus lesions resulted in insomnia. Posterior hypothalamus lesions resulted in sleepiness The arrow indicates lesions resulting in narcolepsy. These observations lead to hypothesis of a "sleep center" in anterior hypothalamus and a "wake center" the posterior hypothalamus.
question
Recording sleep physiology
answer
Wake and sleep states have distinguishing physiological characteristics that can be measured by polysomnography: EEG (electorencephalogram: cortical activity measured by electrodes on the scalp), EOG (electrooculogram: eye movement measured by electrodes adjacent to the eyes), EMG (electromyogram: muscle tone measured by electrodes picking up chin muscle activity) as well as by behavioral characteristics
question
Sleep/wake stages: wake, REM, NREM (stage 1-4)
answer
Awake: high frequency and low amplitude Stage 1 - Stage 4: Frequency continues to decrease and amplitude increases REM Sleep: Similar waves to Awake; high brain activity
question
Sleep patterns
answer
Patterns across the sleep cycle: Typical adult pattern of these different stages during the night NREM predominates early with cycling between light (stage 1) and deep (stage 4) sleep REM episodes increase in freq and duration later in the night and is accompanied by physiological changes (incr EOG, heart, resp; decr EMG)
question
Sleep patterns across the life cycle (1)
answer
Sleep patterns across the life cycle (1)
question
Toddler sleep

answer
Rapid descent at bedtime into deep NREM sleep, awakenings throughout the night
question
Adolescent sleep
answer
Difficulty falling asleep and waking up at prescribed times
question
Older adult sleep
answer
Awakenings throughout the night, less time spent in deep NREM sleep
question
Distribution of sleep stages across the life cycle
answer
Relative proportion of time spent in different stages of sleep also changes across the life cycle: Although the timing of these sleep stage changes in utero is not known with certainty (dotted lines), data from premature infants are consistent with REM sleep occupying most of life at a gestational age of 26 weeks. After 26 weeks, the time spent in waking increases until death. Infants spend equal time in wake/REM/NREM sleep (REM is 50% of sleep) By adulthood, REM has declined significantly (REM is 25% of sleep) NREM is relatively constant
question
Fragmentation of sleep-wake cycle with aging
answer
Younger man shows high daytime activity and extended rest periods at night Older man shows more fragmentation of the sleep/wake cycle: more naps in day, more awakenings in night
question
Wake pathways: ascending reticular formation in brainstem and posterior hypothalamus (aka reticular activating system)
answer
Cholinergic (ACh) neurons in basal forebrain and pons [pedunculopontine/lateral dorsal tegmental areas (PPT/LDT)] activate the cortex Locus coeruleus (LC) (norepinephrine; NE), raphe nuclei (serotonin; 5-HT), tuberomamillary nucleus (TMN) (histamine; HA), substantia nigra/ventral tegmental area (SN/VTA) (dopamine; DA) promote wakefulness via cortical and subcortical projections Orexin/hypocretin neurons in posterior hypothalamus innervate arousal systems and cortex Wakefulness is a complex behavioral state representing the coordinated expression of many individual behaviors that are constantly changing in response to internal/external stimuli. As a result, there are many neurotransmitters, peptides and brain regions that are part of the 'arousal network'. Not every circuit is activated at every moment of the wake cycle: some may be engaged under select conditions i.e. during stress, during periods of physical activity, etc). Ex 1: NE system promotes arousal during stress or cognitive challenge for optimal attention and task performance Ex 2: DA system promotes arousal during conditions of high motivation or physical activity Ex 3: orexin system sustains arousal and promotes arousal responses to homeostatic challenge (driving motivated behaviors like food seeking) In general, neurons that are part of the arousal network meet the following criteria: neuronal activity and neurotransmitter release is highest during the wake cycle and lowest in the sleep cycle treatments that enhance signaling (i.e. agonists) promote physiological and behavioral indicators of wakefulness treatments that inhibit signaling (i.e. antagonists, lesions, disease) promote physiological and behavioral indicators of sleep Several major neurotransmitter and neuropeptide systems that promote wakefulness reside in the brainstem reticular formation and the posterior hypothalamus. These include the cholinergic, aminergic (panel A) and hypocretin/orexin systems (panel B). Acetylcholine (ACh): one group of cells is located in the pontine reticular nuclei (pedunculopontine and laterodorsal tegmental area) another group in basal forebrain Aminergic systems Norepinephrine (NE): cells located in pontine reticular formation (locus coeruleus) Serotonin (5-HT): cells located in the midbrain reticular formation (raphe nuclei) Histamine (HA): cells located in posterior hypothalamus (tuberomamillary nucleus) Dopamine (DA): cells located in midbrain reticular formation (ventral tegmental area and substantia nigra) Orexin/hypocretin: cells located in posterior hypothalamus; Projections: other areas of the arousal network in the brainstem reticular formation and post. hypothalamus SUMMARY: Wakefulness is the coordinated product of an arousal network that includes cholinergic, aminergic and peptidergic systems residing in the brainstem reticular formation and posterior hypothalamus
question
NREM sleep pathways
answer
GABA/galanin ventrolateral preoptic area (VLPO) neurons in anterior hypothalamus inhibit arousal systems in NREM sleep. NREM sleep: GABA (shown in pink) Location: anterior hypothalamus (ventrolateral preoptic area; VLPO) Projections: the arousal network in the brainstem reticular formation and posterior hypothalamus These anterior hypothalamus neurons show the greatest neuronal activity and neurotransmitter release during NREM sleep; treatments that enhance GABA signaling (ex: benzodiazepines such as Valium; non BZ GABA agonist hypnotics such as Lunesta, Sonata, Ambien) induce sleep SUMMARY: NREM sleep results from descending inhibition of the arousal network by inhibitory GABAergic neurons of the anterior hypothalamus
question
NREM control
answer
VLPO neurons (orange) in anterior hypothalamus are activated in NREM sleep VLPO neurons are GABAergic and inhibit neurons in the arousal system (such as the histamine neurons in the TMN of the posterior hypo-thalamus) (pink)
question
REM sleep pathways
answer
Cholinergic 'REM-on' PPT/LDT neurons are inhibited by a subpopulation of 'REM-off' 5-HT/NE/histamine neurons in wake and to a lesser extent in NREM but totally released from inhibition only during REM Cholinergic PPT/LDT neurons drive thalamocortical desynchrony in REM (via thalamus projections) and drive atonia in REM (via projections to medial medulla neurons that inhibit motor output) REM sleep Acetylcholine Location: a specific population of 'REM-on' neurons resides in the pontine reticular formation (pedunculopontine and laterodorsal tegmental area) Projections: thalamus, medial medulla Thalamus projecting REM-on neurons drive thalamocortical desynchrony in REM Medial medulla projecting REM-on neurons contribute to atonia in REM REM-on neurons show the greatest neuronal activity and neurotransmitter release during REM sleep; treatments that enhance ACh signaling stimulate REM whereas treatments that inhibit ACh signaling reduce REM REM-on neurons are normally under inhibitory control by aminergic pathways (such as 5-HT, NE, HA; REM-off cells) during wake and also during early NREM sleep but by the end of NREM episodes, this subset of inhibitory neurons is also shut off by the VLPO/GABA sleep circuit, thus REM-on neurons are disinhibited and initiate a REM episode. SUMMARY: REM sleep results from disinhibition of a specific population of 'REM-on' cholinergic neurons in the pontine reticular formation; ascending projections drive cortical desynchrony and descending projections drive atonia during REM
question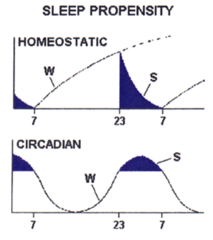
Sleep Regulation

answer
Homeostasis: - "Sleep pressure" accumulates during wake and recovers during sleep - Chemical basis: somnogen - Challenges (sleep deprivation or naps) are followed by recovery sleep in proportion to the challenge Circadian rhythms: 24 h cycle of sleep/wake Sleep is regulated by different interacting processes, individually modeled in the two panels: Homeostasis: "sleep pressure": system recovers to an optimal set point when perturbed: challenges (ex: sleep deprivation, naps) are followed by recovery sleep in proportion to the challenge (ex: equal to sleep loss, gain); clearly this implies that there is an optimal level of sleep and a physiological "need" for to maintain that level; mechanism: some substance accumulates during prolonged wakefulness that drives the "need" for sleep (somnogen) Circadian rhythms: 24 h cycle of sleep/wake These processes interact to result in the normal sleep/wake cycle
question
Adenosine as a somnogen
answer
Adenosine is a good candidate for a somnogen Induces sleep Levels vary with sleep-wake cycle Criteria #1: sleep is increased and wake reduced by injection of adenosine or adenosine agonists into basal forebrain (upper panel A), anterior hypothalamus or the reticular formation (upper panel B) Criteria #2: adenosine levels highest in wake, lowest in sleep (lower panel A); breakdown product of ATP; during prolonged wakefulness, energy producing systems run down (ATP levels are depleted as they are broken down into ADP, AMP and eventually adenosine): this causes rise in adenosine levels in areas of the brain like the basal forebrain (lower panel B)
question
Adenosine and NREM Circuits
answer
Adenosine promotes sleep by activating NREM circuits. Mechanism of action: adenosine promotes sleep by activating NREM circuits Falling asleep: Adenosine builds up during the day and stimulates the activity of VLPO sleep-promoting cells which in turn inhibit brain regions involved in arousal (such as histamine, orexin cells in posterior hypothalamus and 5-HT and NE cells in brainstem) Waking up: Light signals are received by SCN in the hypothalamus via a retinohypothalamic tract: 1. shutting down VLPO neurons and disinhibiting the arousal network 2. stimulating orexin neurons which also stimulate the arousal network
question
Homeostatic sleep challenges
answer
Sleep deprivation (increase in sleep pressure) increases NREM and REM sleep Naps (reduction of sleep pressure) decrease NREM sleep Sleep regulation: homeostasis (demonstrated by the effects of sleep deprivation) Experimental elevation of sleep pressure: sleep deprivation (24 h) in rat: NREM (delta) increases: higher peaks in delta shows the presence of very large, slow waves; recovery is in proportion to sleep loss REM: episode duration increases and episode frequency increases (REM rebound) Experimental reduction of sleep pressure: naps Napping produces a corresponding decrease in sleep in the post-nap night, and furthermore, shown here, naps that contain large amounts of NREM sleep have a bigger effect on the postnap night (in terms of reduced NREM sleep) than do naps that contain smaller amounts of NREM sleep (NREM response is in proportion to the NREM gain produced by the nap) There are homeostatic mechanisms for both NREM and REM sleep
question
Sleep Functions
answer
Physiological restorative functions, energy conservation Memory consolidation - Memory traces are encoded in hippocampus (short-term) and are gradually stabilized by repeated reactivation of the memory trace for long-term storage - Sleep is a time when memories can be reactivated and reconsolidated in the absence of new sensory input - Dream content is in part the "conscious" experience of these reactivation/reconsolidation processes - Evidence: - New learning and particularly salient daytime experiences are represented in dream content as a way to repeat the experience - Sleep following learning facilitates consolidation and improves recall of newly learned material - Sleep deprivation (both REM and NREM) impairs consolidation and recall of newly learned material Homeostatic drive for sleep indicates its importance Many theories about why it's important including its restorative and energy conserving features Memory consolidation is another theory for which there is increasingly abundant evidence
question
Circadian rhythms: behavior, physiology and hormones
answer
In addition to homeostatic regulation of sleep, there are circadian processes as well Circadian literally means 'about a day' Daily rhythms in alertness, body temperature, growth hormone and cortisol secretion, and potassium excretion Also hormonal rhythms such as melatonin secretion from the pineal gland Daily rhythms in rest-activity, computation speed (number of computations performed per minute), and time estimation (accuracy with which short intervals of time are assessed);
question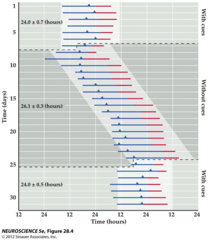
Synchronized vs. desynchronized 'free running' sleep-wake cycle

answer
Normally, our sleep-wake cycle follows a 24 h rhythm that is synchronized with the 24h day Endogenous rhythmicity of the sleep-wake cycle can only be assessed under conditions of isolation from time cues (constant darkness, constant temperature, no clocks, TVs etc) With external cues (light, temperature, clocks), sleep/wake follows a 24 schedule; without those cues (constant conditions), subjects 'free run': humans have a period closer to 25 h: they wake up about 1 hour later each day under these conditions. Since rhythms persist in the absence of external time cues, this is evidence that rhythms are generated from an internal pacemaker that has a period of about 25 hrs in humans Entrainment is the process whereby external time cues (i.e. light) synchronize the pacemaker to the 24 h day
question
Model of the circadian system
answer
A circadian system can be modeled as having three components: an input pathway by which environmental information synchronizes the oscillator, a set of elements which feedback on one another to form the oscillator, an output pathway by which the oscillator is coupled to the processes it controls. What brain region or regions fulfills the criteria of oscillator? Must have intrinsic oscillatory activity Must have the ability to synchronize its activity with the external environment Must drive all the rhythmic outputs of the organism
question
Entrainment pathway
answer
Retina: A specific population of retinal ganglion cells project directly to the SCN (via the retinohypothalamic tract; RHT). The RHT is part of the optic nerve until the optic chiasm where it separates from other optic fibers to project to the SCN. RHT terminals release glutamate in the SCN which stimulate SCN neurons, synchronizing the oscillations
question
What does an SCN lesion do?
answer
What is the evidence that the SCN is the locus of intrinsic oscillatory activity? SCN lesion results in loss of circadian function. This is a record of activity of an rat maintained in a light-dark cycle. From the top of the record to the arrow, the animal exhibits a normal rhythm of activity, indicated by dark areas. The record is double plotted, which means that each line shows the preceding day and the new day to ease evaluation of the record. At the arrow, a bilateral SCN lesion was performed. Activity is distributed randomly thereafter, meaning circadian organization of rest-activity has been lost. Rhythmicity can be restored in these lesioned animals by transplantation of SCN from another animal
question
SCN: intrinsic activity
answer
SCN cells show circadian variation of cellular metabolism (glucose utilization), vasopressin release and of neuronal activity in vivo even when the SCN is surgically isolated (knife cuts to remove all inputs) or in SCN brain slices or in dissociated SCN cells All of these SCN properties demonstrate that oscillation is intrinsic to the SCN: they can operate without inputs from other brain regions, etc.
question
SCN Outputs
answer
Primary projections of SCN are limited but secondary projections are widespread and likely responsible for the wide range of behavioral, physiological and hormonal circadian rhythms
question
Circadian pathway: inputs and outputs
answer
1. Light stimulates the SCN via glutamate release from retinal ganglion cells of the RHT 2. SCN cells release inhibitory neurotransmitter GABA which ultimately inhibits melatonin secretion from the pineal gland 3. Therefore, melatonin is secreted at night This diagram illustrates SCN outputs as well as inputs and shows how it controls rhythmic secretion of melatonin from the pineal gland Light stimulates the SCN through excitatory glutamate release from retinal ganglion cells of the RHT. SCN cells then release the inhibitory neurotransmitter GABA which ultimately inhibits secretion of melatonin from the pineal gland. This means that melatonin secretion occurs at night. Melatonin also feeds back onto the SCN and can itself cause phase shifts in SCN oscillations. Abbreviations: IML, intermediolateral cell column of the spinal cord; Mel, melatonin; PVN, paraventricular nucleus; RHT, retinohypothalamic tract; SCG, superior cervical ganglion; SCN, suprachiasmatic nucleus.
question
Disruption of circadian rhythms
answer
Entrainment stimuli at the wrong time of the sleep/wake cycle Ex: light pulse early in subjective night produces a phase delay (subject wakes up later than normal) Ex: light pulse late in subjective night produces a phase advance (subject wakes up earlier than normal) Melatonin also produces predictable phase shifts If entrainment stimuli such as light shown here are given at the wrong time in the day-night cycle, such as a light pulse in the middle of the night, they can shift the phase of the circadian oscillator in predictable ways If a light pulse is introduced early in subjective night, a phase delay occurs: subject wakes up later than normal If a light pulse is introduced late in subjective night, a phase advance occurs: subject wakes up earlier than normal Melatonin is normally secreted at night - its circadian release is driven by the SCN but it also feeds back on the SCN; if taken as medication, it can produce phase shifts If administered in late subjective night - it will produce a phase delay (opposite of the effect of light at this time) If administered in subjective morning - it will produce a phase advance Phase shifting can be used as therapies for desynchronization problems (shift work, jet lag): timed light exposure or melatonin administration to reset the internal clock to synchronize with the external world
question
Disruption of circadian rhythms
answer
This kind of phase shifting occurs under a number of circumstance that many of us encounter on a regular basis Desynchronization of the internal clock from external cues can be very dysfunctional Dysfunction of circadian rhythms: Desynchronization of the internal clock from external cues can occur in two major circumstances: Shift work (professions requiring constant schedule change) and jet lag (rapid movement across time zones) The consequences of these circumstances is loss of sleep, fatigue, stomach ailments and compromised performance, both cognitive and motor
question
Effects of Reducing Interns' Work Hours on Serious Medical Errors in Intensive Care Units
answer
Interns made substantially more serious medical errors when they worked frequent shifts of 24 hours or more than when they worked shorter shifts. Eliminating extended work shifts and reducing the number of hours interns work per week can reduce serious medical errors in the intensive care unit. Shift work of medical interns in an ICU: traditional schedule = constantly changing schedules and long shifts (28h) Consequences: medical errors Treatment: intervention schedule that cuts longest shift time in half (14h) - results in significantly fewer serious errors such as diagnostic and medication errors
question
Drugs that affect sleep/wake cycle
answer
Sleep altering drugs (SSRI, tricyclic ADs, stimulants, benzodiazepines, antihistamines, typical antipsychotics): while some of these drugs are used specifically to alter the sleep wake cycle (ex: benzodiazepines or sedatives to treat insomnia), many of the drugs on this list are used to treat other disorders and their sleep-altering properties are considered side-effects Notice that all of these drugs act on the many neurotransmitter systems that I described earlier as modulators of either sleep-wake cycle or circadian rhythms: 5-HT, NE, DA, histamine, GABA
question
Drugs and sleep patterns
answer
Benzodiazepines stimulate GABA signaling Caffeine is an adenosine receptor antagonist
question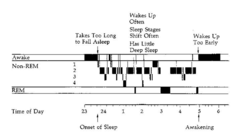
Insomnia

answer
An overview of some of the more common sleep disorders: Insomnia: this patient has trouble falling asleep and staying asleep Insomnia is defined as an inability to obtain sleep of sufficient length/quality to feel rested the next day, affects 40% of women, 30% of men, tends to increase with age; insomniacs may be in a constant state of hyperarousal Short-term treatment with benzodiazepines; changing sleep habits
question
Parasomnias
answer
Sleep walking: mix of wake and NREM sleep Sleep terrors: mix of wake and NREM sleep REM sleep behavior disorder: mix of wake and REM sleep Parasomnias : behaviors normally associated with wakefulness occur during sleep (mixing of sleep and wake stages) Sleep walking: common in children (1-17%), peaks at 11-12 yrs; occurs in stage IV sleep Sleep terrors: disorder of arousal; person is in stage 3/4 NREM sleep but sleep terror is initiated by a scream and severe panic followed by prominent motor activity (hitting the wall, running around the room, out of the house), universal feature is inconsolability REM sleep behavior disorder: 90% of those affected are men, usually over 50 yrs of age, failure of atonia in REM; vigorous motor behaviors accompanying vivid dreams - people 'act out' their dreams; acutely RBD can be a side effect of drugs such as SSRIs, chronic RBD is sometimes associated with neurological disorders (Parkinson's and other neurodegenerative disorders); effectively treated with benzodiazepines
question
Narcolepsy
answer
Fragmented sleep/wake patterns Excessive daytime sleepiness Sometimes cataplexy (loss of muscle tone during emotional situations) REM abnormalities: hallucinations at sleep onset/offset, sleep paralysis (waking to temporary paralysis) This rest-activity pattern of a narcoleptic patient shows that day/night patterns are not very different: less rest at night, less activity in day
question
Loss of orexin/hypocretin cells in narcolepsy
answer
Hypocretin/orexin cell loss, possibly the target of an autoimmune insult; HC cells project to and provide excitatory drive to histamine, dopamine, norepinephrine, 5-HT and ACh cells of the arousal network Since HC promotes wakefulness, opposing rising sleep drive, loss of these cells may account for excessive daytime sleepiness and 'sleep attacks' Also, 90% of narcoleptics w/cataplexy have low/undetectable HC levels in CSF - useful diagnostic tool Treatments include naps, regular sleep schedule, stimulants like methylphenidate (Ritalin) or modafinil; SSRIs or tricyclic antidepressants for cataplexy
question
Sleep apnea
answer
Sleep apnea: interrupted breathing during sleep, associated with fat build up or loss of muscle tone with aging: upper airway collapses when muscles relax during sleep, often associated with loud snoring; when breathing stops, blood oxygen falls and triggers an awakening and the restoration of airway muscle tone; this cycle can be repeated up to 100 times/hr leading to major sleep loss Treatments include weight loss, changing sleep position, corrective devices, surgery. This intermittent pattern of oxygen loss has been modeled in animals and has been shown to be neurotoxic in hippocampus, an area critical for learning and memory. This kind of cell loss could add to the cognitive impairment that results from sleep loss.
question
Restless legs syndrome
answer
Representative patient descriptions of RLS sensations in the legs • Like an electrical current • Like Coca Cola bubbling through my veins • The "gotta moves" • Aching in my bones • Like maggots crawling through my limbs • Creepy crawly • Throbbing • Like a toothache in the legs • The "heeby-jeebies" • Crazy legs • "Jimmy" legs • Painful • Pulling • Tearing • Itching bones • Growing pains Restless legs syndrome: unpleasant tingling sensations in legs and feet and an urge to move them for relief; common especially in older people (accounts for 1/3 of the insomnia in people older than 60 yrs); constant leg movement in day and fragmented sleep/insomnia at night Treated with anti-parkinsonian agents, benzodiazepines, opiates, anti-convulsants
question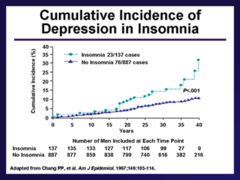
Psychiatric disorders with associated sleep disturbance

answer
Major depressive disorder Seasonal affective disorder Anxiety disorders Post-traumatic stress disorder Schizophrenia Sleep dysfunction: associations with other psychiatric disorders Over 90% of patients with major depression have sleep disturbances (insomnia, sleep maintenance difficulty, early morning awakenings); depressed patients also have disrupted circadian rhythms (sleep/wake, cortisol, melatonin secretion, body temperature); since depression is associated with 5-HT dysfunction, disruption of the serotonergic input to SCN may contribute to circadian dysfunction and sleep disturbance The picture shows the cumulative incidence of depression in 1,045 men according to sleep complaints in medical school: insomnia (•) versus no insomnia (A). The survey was filled out by Johns Hopkins medical graduates. Those individuals who had problems with insomnia or whose sleep was disrupted by stress in med school as they were followed over the years, there's a point of divergence. And the longer that they are followed, the greater the probability that depression will develop, and the sole determining factor here was insomnia versus no insomnia. Seasonal affective disorder: recurrent cycles of fall/winter depression; treatments include SSRIs and phototherapy: bright light exposure in early morning (clinical benefit come from suppression of melatonin secretion) 38% of patients with anxiety disorders have insomnia; posttraumatic stress disorder patients report insomnia as well as nightmares Schizophrenics commonly report insomnia; polysomnography shows increased sleep fragmentation, decreased REM latency, reduced NREM sleep



